The first zinc-catalyzed oxidation of sulfides to sulfones using H2O2 as green oxidant†
Abstract
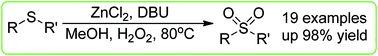
An interesting procedure for the oxidation of sulfides to sulfones has been developed. By using a catalytic amount of zinc salt as the catalyst, DBU as the ligand, various sulfones were produced in good yields with hydrogen peroxide as the terminal green oxidant. Note that this is the first example of zinc-catalyzed oxidation of sulfides to sulfones.

 Please wait while we load your content...
Please wait while we load your content...