Vibrational study of adsorption of Congo red onto TiO2 and the LSPR effect on its photocatalytic degradation process†
Abstract
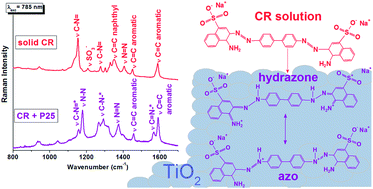
Chemical interaction between the azo dye Congo Red (CR) and two photocatalysts based on titanium dioxide (commercial Degussa P25 TiO2 and nanostructured TiO2, primarily in the form of anatase) were analyzed by diffuse reflectance and resonance Raman spectroscopies. The adsorption isotherms of the dye over the photocatalysts were consistent with the Langmuir adsorption model. High similarity between the CR dye adsorbed onto P25 TiO2 and the product of CR oxidation/acidification (hydrazone tautomer) was observed through resonance Raman spectroscopy. The vibrational results were consistent with the analysis of the Langmuir isotherm, where chemisorption of CR on P25 and physisorption of CR on nanoTiO2 were in accordance with the obtained Kads and qmax. The addition of plasmonic Ag NPs was performed in order to analyze effects on TiO2 photocatalytic efficiency. The CR photodegradation with the plasmonic catalyst presented an increase of kobs of 14% and 100% compared to P25 and nanoTiO2, respectively. The catalysis improvement was assigned to the near field enhancement due to the Ag NPs plasmonic effect.

 Please wait while we load your content...
Please wait while we load your content...