Self-radiolysis of tritiated water. 2. Density dependence of the yields of primary species formed in the radiolysis of supercritical water by tritium β-particles at 400 °C
Abstract
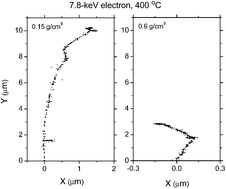
Supercritical water (SCW) has attracted increasing attention after the Generation IV International Forum selected the supercritical water-cooled reactor (SCWR) as one of six concepts for further investigation. The reference design for the SCWR calls for an operating pressure of 25 MPa and a core outlet temperature as high as 625 °C. Tritium is of special interest in these proposed systems, because of the appreciable quantities that would be produced. Regarding the water chemistry in SCWR systems, there is however a complete lack of information on the radiolysis of SCW by tritium β-particles. Because direct measurement of the chemistry under such extreme conditions of high temperature, pressure, and mixed neutron and β/γ radiation fields is difficult, chemical models and computer simulations are important for predicting the detailed radiation chemistry of the cooling water in a SCWR core and the impact on materials. In this study, Monte Carlo simulations were used to predict the yields (or G-values) for the primary species e−aq, H˙, H2, ˙OH, and H2O2 formed from the radiolysis of deaerated SCW (H2O) by the low-energy β-electrons (∼18.6 keV maximum) of tritium at 400 °C as a function of water density in the range of ∼0.15–0.6 g cm−3 (∼24–56 MPa). The objective was to elucidate the (time-dependent) mechanisms involved in the self-radiolysis of tritiated water under supercritical conditions. Calculated yields were compared with data obtained for low-“linear energy transfer” (LET) radiation (such as 60Co γ-rays or high-energy electrons) and fast neutrons. Our simulations revealed that there was a strong resemblance between the density dependences of the different yields for the radiolysis of SCW with tritium β− particles and fast neutrons, corroborating very well with a model of tritium β radiolysis mainly driven by the chemical action of “short tracks” of high local LET. As for the effect of density on the various yields, there was an increased “cage” escape of free radicals at low-density SCW. In contrast, these density effects acted in the opposite sense in the high-density liquid-like region where the caged free radical products were forced to remain as colliding neighbors and recombine, thereby increasing the molecular yields. Finally, the occurrence of the reaction of H˙ atoms with water in the homogeneous chemical stage was found to play a critical role in the formation yields of H2 and ˙OH at 400 °C. Recent work has recognized the potential importance of this reaction above 200 °C, but its rate constant is still not well known.

 Please wait while we load your content...
Please wait while we load your content...