Solution plasma synthesis process of tungsten carbide on N-doped carbon nanocomposite with enhanced catalytic ORR activity and durability†
Abstract
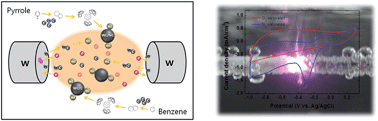
In this study, the enhancement of ORR activity and durability by an N-doped carbon nanocomposite on tungsten carbide (WC) nanoparticles was reported. The nanocomposite of tungsten carbide on two different carbon matrices, pure carbon matrix (WC/C) and N-doped carbon matrix (WC/N–C), was at first prepared by a simple discharge process in the mixture of benzene/dodecane and pyrrole/dodecane. The nanoparticles of tungsten carbide were formed via the sputtering effect of tungsten electrodes during discharge. The results of TEM and XRD demonstrated that tungsten carbide nanoparticles with a mean size of 6 nm were evenly dispersed on both carbon matrices. The results of cyclic voltammetry measurements showed that both obtained metal/carbon matrices promoted a significant oxygen reduction reaction (ORR) in alkaline solution. The ORR potential of tungsten carbide/carbon matrix and nitrogen-doped carbon were −0.29 V and −0.36 V, respectively. The enhancement of ORR activity in WC/N–C was attributed to the combined catalytic effects of WC and N in the carbon matrix. Although the ORR activity of WC/N–C was still incomparable with commercial Pt/C, the durability of the catalyst was significant higher than that of Pt/C in a methanol environment. The catalyst did not exhibit an evident change of initial current after 4000 s. Therefore, the inexpensive N-doped WC/C nanocomposite might be a promising and highly durable catalytic material for cathodes in fuel cell applications.

 Please wait while we load your content...
Please wait while we load your content...