Three dimensionally ordered macroporous Au/CeO2 catalysts synthesized via different methods for enhanced CO preferential oxidation in H2-rich gases†
Abstract
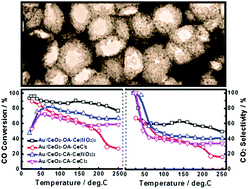
Three dimensionally ordered macroporous (3DOM) Au/CeO2 catalysts were synthesized via a thermal decomposition-assisted colloidal crystal templating method following different synthetic procedures using citric acid and oxalic acid as chelating ligands and CeCl3 and Ce(NO3)3 as cerium salt precursors. All 3DOM Au/CeO2 catalysts possess well-defined 3DOM skeletons composed of well-crystallized CeO2 nanoparticles, based on the different synthetic procedures, the 3DOM Au/CeO2 catalysts show variations in surface elemental compositions, particle sizes of CeO2, macroporous and mesoporous structures, and valence states of Au. The mesoporous walls with nanopores ∼3–4 nm on 3DOM Au/CeO2 skeletons can be created when using Ce(NO3)3 as precursor. The catalytic performance of 3DOM Au/CeO2 catalysts for CO preferential oxidation in H2-rich gases was systematically investigated. The catalytic performance is closely correlated to surface elemental compositions, particle sizes of CeO2, macroporous and mesoporous structures, and valence states of Au due to the different synthetic procedures. The 3DOM Au/CeO2 catalysts prepared using oxalic acid as chelating ligand and Ce(NO3)3 as cerium salt precursor show the highest catalytic activity for CO preferential oxidation in H2-rich gases with 90.1% CO conversion and 59.9% CO2 selectivity at 50 °C, and 88.3% CO conversion and 59.3% CO2 selectivity at 80 °C, respectively. The obtained 3DOM Au/CeO2 catalysts may be of importance for guiding the design of efficient catalysts with desired porous structures that are potentially applicable in polymer electrolyte membrane fuel cells.

 Please wait while we load your content...
Please wait while we load your content...