Expanding the scaffold for bacterial RNA polymerase inhibitors: design, synthesis and structure–activity relationships of ureido-heterocyclic-carboxylic acids
Abstract
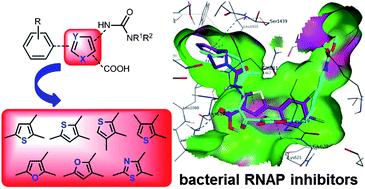
The emergence of bacterial resistance requires the development of new antibiotics with an alternative mode of action. Based on class I, developed in our previous study, a new series of RNA polymerase (RNAP) inhibitors targeting the switch region was designed. Feasible synthetic procedures for the aryl-ureido-heterocyclic-carboxylic acids were developed including three regioisomeric thiophene classes (II–IV), as well as three isosteric furan (V, VI) and thiazole (VII) classes. Biological evaluation using a RNAP transcription inhibition assay revealed that class II compounds possess the same activity as the parent class I, whereas classes III, V–VII were active, however with lower potency. Structure–activity relationship (SAR) studies, supported by molecular modeling, elucidated the structural requirements necessary for interaction with the binding site. Beside the RNAP inhibitory effects, the new compounds displayed good antibacterial activities against Gram positive bacteria and the Gram negative E. coli TolC strain. Moreover, they showed no cross resistance with the clinically used RNAP inhibitor rifampicin (Rif) and a lower rate of resistance compared to Rif.

 Please wait while we load your content...
Please wait while we load your content...