Electrochemical treatment of reverse osmosis concentrate generated by the leather industry using a Cu–graphite electrode†
Abstract
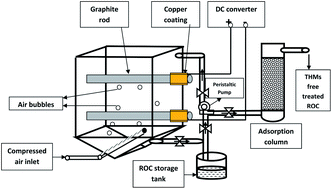
Electrochemical treatment of reverse osmosis concentrate (ROC) generated by the leather industry was evaluated using a copper coated graphite (Cu–graphite) electrode for the elimination of refractory organic pollutants. The effect of dilution of the ROC and applied current density on the performance of electrochemical oxidation was investigated. The presence of chloride ions in the ROC favoured the complete elimination of TKN and COD removal by 98% at optimum conditions: current density, 100 mA cm−2 and electrolysis period, 6 h. The increase in ROC dilution slightly decreased the percentage removal of COD from 98% to 96%. The effect of current density on the removal of COD and TKN in raw ROC was found to be lower than in more diluted ROC. It was concluded that electrochemical treatment was more favourable for ROC samples without dilution. The generation of trihalomethanes during the electrochemical oxidation of ROC was effectively removed in an activated carbon packed bed column.

 Please wait while we load your content...
Please wait while we load your content...