Vinyl acetate living radical polymerization mediated by cobalt porphyrins: kinetic–mechanistic studies†
Abstract
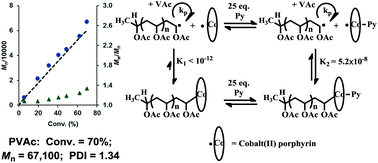
Radical polymerization of vinyl acetate (VAc) in the presence of cobalt(II) tetramesitylporphyrin, CoII(TMP), as a mediator was performed in a series of solvent environments. Cobalt porphyrin was observed to mediate an effective living radical polymerization reaction of vinyl acetate in bulk as shown by the linear increase in molecular weight with conversion, relatively narrow molecular weight distributions (Mw/Mn = 1.24–1.34), and molecular weights close to the theoretical value up to moderately high monomer conversions (∼70%). The levels of deviation for the observed PVAc molecular weights from the theoretical values varied with the choice of solvent medium, which suggests that the control of vinyl acetate radical polymerization was affected by chain transfer to the solvent. Non-coordinating solvents influence the polymerization rate primarily by different propagation rate constants (kp). Pyridine coordination with dormant organo-cobalt(III) increases the dissociation constant and changes the dominant polymerization process from degenerative transfer to reversible termination.

 Please wait while we load your content...
Please wait while we load your content...