Redox-responsive core cross-linked micelles based on cypate and cisplatin prodrugs-conjugated block copolymers for synergistic photothermal–chemotherapy of cancer†
Abstract
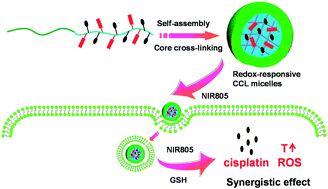
Responsive cross-linked block copolymer micelles which have emerged as promising drug delivery systems showed high stability and on-demand drug release. The combination therapy of cancer can be achieved via co-delivery of varying therapeutic molecules in one system. Here, we developed a redox-responsive core cross-linked (CCL) micelle conjugated with cypate and cisplatin prodrugs within the cores for synergistic photothermal and chemotherapy. The block copolymer, poly[(2-(2-methoxyethoxy)ethyl methacrylate)-co-(N-methacryloxy succinimide)]-block-poly(N-(2-hydroxypropyl) methacrylamide) (P(MEO2MA-co-MASI)-b-PHPMA), was synthesized via reversible addition–fragment chain transfer (RAFT) polymerization. After partial amidation reaction of succinimide with 3-azidopropylamine, the alkynyl-functionalized cypate and Pt(IV) complex were conjugated via click reaction. The CCL micelles were fabricated by core cross-linking at 37 °C in aqueous solution using cystamine as the cross-linker. P(Pt-Cy-MEO2MA)-b-PHPMA CCL micelles showed redox-responsive cross-linker cleavage and cisplatin drug release in the presence of reductants. The conjugated cypate moieties in the cores of CCL micelles resulted in photothermal temperature increase and reactive oxygen species (ROS) generation under 805 nm near infrared (NIR) laser irradiation. The cytotoxicity of CCL micelles was investigated with and without NIR irradiation. A significant synergistic effect of photothermal therapy and chemotherapy was demonstrated against cisplatin-resistant human lung cancer cells A549R under NIR irradiation.
- This article is part of the themed collection: Celebrating The Five-Year Anniversary of Polymer Chemistry

 Please wait while we load your content...
Please wait while we load your content...