Improved photovoltaic properties of terpolymers containing diketopyrrolopyrrole and an isoindigo side chain
Abstract
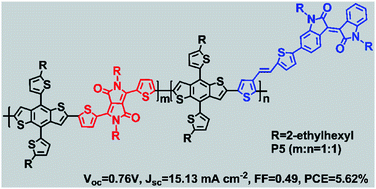
A series of donor–acceptor (D–A) conjugated random terpolymers were synthesized by copolymerizing electron-rich alkylthienyl-substituted benzodithiophene (BDTT) and two electron-deficient units, a diketopyrrolopyrrole (DPP) moiety and isoindigo (TID) based side chain. The effects of the DPP and TID units on the thermal, photophysical and electrochemical properties of the polymers were investigated using thermogravimetric analysis, UV-vis-NIR absorption spectra, and cyclic voltammetry. Compared with the parent polymers (P1 and P2), the optical properties of the random terpolymers (P3–P6) were controlled successfully by tuning the ratio of DPP and TID. The increase in TID content induced an increased absorption between 450 and 600 nm and a lower highest occupied molecular orbital (HOMO) energy level, while higher DPP content resulted in stronger absorption between 600 and 900 nm. Bulk heterojunction solar cells based on the as-synthesized polymers as electron donors and (6,6)-phenyl-C-butyric acid methyl ester (PCBM) as the acceptor were fabricated. The best power conversion efficiency (PCE) of 5.62% was obtained from P5 (DPP : TID = 1 : 1) due to its high short-circuit current density (Jsc higher than 15 mA cm−2), mainly arising from the broadened light absorption. The results have demonstrated that the random terpolymers, with complementary light-absorption, have a great potential for increasing the photocurrent and PCE in polymer solar cells.

 Please wait while we load your content...
Please wait while we load your content...