Chain transfer to solvent in the radical polymerization of structurally diverse acrylamide monomers using straight-chain and branched alcohols as solvents†
Abstract
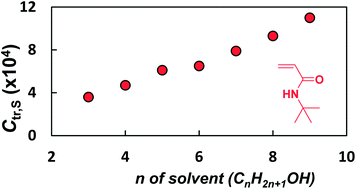
Chain transfer to solvent in conventional radical polymerizations of N-tert-butylacrylamide (TBAM) and N-(2-morpholin-4-ylethyl)acrylamide (MEA) in a range of alcohol solvents is investigated. Mayo analysis of polymerization of TBAM in linear alcohols (C3–C9) resulted in an approximately linear increase in chain transfer to solvent constant (Ctr,S) with the number of methylene (CH2) units in the solvent. The branched alcohol 3-methyl-3-pentanol gave the smallest Ctr,S (using Mayo analysis), and thus allowed attainment of higher molecular weights (MWs) in the nitroxide-mediated polymerizations (NMP) of TBAM. Overall, the data show that MEA is more prone to chain transfer to solvent than TBAM (higher Ctr,S), and further analysis of the conventional radical polymerization of MEA in 3-methyl-3-pentanol indicate chain transfer to monomer may also be occurring. The first controlled/living polymerizations of MEA are detailed with chain transfer having a greater impact on maximum achievable MWs in NMP in comparison to TBAM.

 Please wait while we load your content...
Please wait while we load your content...