Abstract
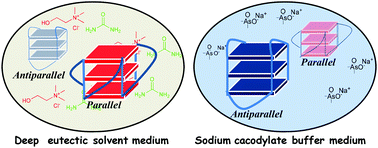
The biocompatibility as well as the sustainability of a deep eutectic solvent makes it a good substitute for aqueous media in studying biomolecules. Understanding the structure and stability of natural and non-natural G-quadruplexes in aqueous and highly viscous media will be useful in biological and nanodevice applications. We report the synthesis and conformational analysis of a model G-rich oligonucleotide G3T3 and non-natural G-rich sequences Pyr1–Pyr3 in aqueous and highly viscous media. Progressive increases in the loop replacement with a non-natural pyrene linker leads to a systematic increase of the thermal denaturation temperature of the modified G-rich oligonucleotides Pyr1–Pyr3 in 10 mM cacodylate buffer (pH 7.2) containing 100 mM KCl, as monitored using UV-Vis spectroscopy. A circular dichroism signal clearly revealed the formation of a predominantly anti-parallel vs. parallel conformation in the natural G-rich oligonucleotide G3T3 as well as the non-natural G-rich oligonucleotides Pyr1–Pyr3 in 10 mM cacodylate buffer (pH 7.2) containing 100 mM KCl. On the other hand, we observed thermodynamic destabilization of G-rich oligonucleotides in a deep eutectic solvent (DES; 1 : 2 choline chloride–urea) containing 100 mm KCl with an increase in loop replacements. Interestingly, we observed an exclusively parallel G-quadruplex conformation in the case of G3T3 in DES containing 100 mm KCl. While pyrene containing G-rich oligonucleotides Pyr1–Pyr3 exhibited a predominantly parallel vs. anti-parallel G-quadruplex conformation in DES containing 100 mM KCl.
- This article is part of the themed collection: Dedicated to the memory of Prof. Nicholas J. Turro

 Please wait while we load your content...
Please wait while we load your content...