Temperature and oxygen-concentration dependence of singlet oxygen production by RuPhen as induced by quasi-continuous excitation
Abstract
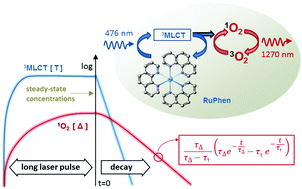
Assessment of partial pressure of oxygen (pO2) by luminescence lifetime measurements of ruthenium coordination complexes has been studied intensively during the last few decades. RuPhen (dichlorotris(1,10-phenanthroline) ruthenium(II) hydrate) is a water soluble molecule that has been tested previously for in vivo pO2 detection. In this work we intended to shed light on the production of singlet oxygen by RuPhen. The quantum yield of singlet oxygen production by RuPhen dissolved in 0.9% aqueous NaCl solution (pH = 6) was measured at physiological temperatures (285–310 K) and various concentrations of molecular oxygen. In order to minimize the bleaching of RuPhen, the samples were excited with low power (<2 mW) laser pulses (20 μs long), created by pulsing a cw laser beam with an acousto-optical modulator. We show that, whereas the RuPhen phosphorescence lifetime decreases rapidly with an increase of temperature (keeping the oxygenation level constant), the quantum yield of singlet oxygen production by RuPhen is almost identical in the temperature range of 285–310 K. For air-saturated conditions at 310 K the measured quantum yield is about 0.25. The depopulation rate constants of the RuPhen 3MLCT (metal-to-ligand charge-transfer) state are determined in the absence and in the presence of oxygen. We determined that the excitation energy for the RuPhen 3MLCT→d–d transition is 49 kJ mol−1 in the 0.9% NaCl solution (pH = 6).

 Please wait while we load your content...
Please wait while we load your content...