Carbonate phosphonium salts as catalysts for the transesterification of dialkyl carbonates with diols. The competition between cyclic carbonates and linear dicarbonate products†
Abstract
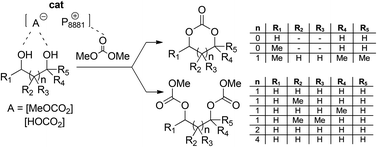
At 90–120 °C, in the presence of methylcarbonate and bicarbonate methyltrioctylphosphonium salts as catalysts ([P8881][A]; [A] = MeOCO2 and HOCO2), the transesterification of non-toxic dimethyl- and diethyl-carbonate (DMC and DEC, respectively) with 1,X-diols (2 ≤ X ≤ 6) proceeds towards the formation of cyclic and linear products. In particular, 1,2-propanediol and ethylene glycol afford propylene- and ethylene-carbonate with selectivity and yields up to 95 and 90%, respectively; while, the reaction of DMC with higher diols such 1,3-butanediol, 2-methyl-1,3-propanediol, 1,3-propanediol, 2,2-dimethyl, 1,3-propanediol, 1,4-butanediol and 1,6-hexanediol produce linear C8–C10 dicarbonates of general formula MeOC(O)O∼∼∼OC(O)OMe as the almost exclusive products. Of note, these dicarbonate derivatives are not otherwise accessible in good yields by other conventional base catalyzed methods. Among 1,3-diols, the only exception was 2-methyl 2,4-pentandiol that yields the corresponding cyclic carbonate, i.e. 4,4,6-trimethyl-1,3-dioxan-2-one. In no one case, polycarbonates are observed. Such remarkable differences of product distributions are ascribed to the structure (branching and relative position of OH groups) of diols and to the role of cooperative (nucleophilic and electrophilic) catalysis which has been proved for onium salts. The investigated carbonate salts are not only effective in amounts as low as 0.5 mol%, but they are highly stable and recyclable.

 Please wait while we load your content...
Please wait while we load your content...