Conjugation-induced fluorescence labelling of mesoporous silica nanoparticles for the sensitive and selective detection of copper ions in aqueous solution†
Abstract
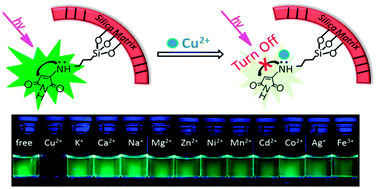
Although the development of sensors for the detection of Cu2+ ions has significant importance, sensitive and selective recognition systems are still limited. Herein, the non-fluorescent small molecule of 2-bromomaleimide (BM) was firstly converted to 2-aminomaleimide (NM) by a simple nucleophilic addition–elimination reaction with pre-grafted amino groups in the meso-space of mesoporous silica nanoparticles (MSNs), and then was designed as fluorescent “on–off” probes for Cu2+ ion recognition. The strong green fluorescence emitted from the pendent NM was induced by the interaction between the lone electron pairs of the N atoms in amino groups and the conjugated maleimide rings. The fluorescence of MSNs labelled NM (MSNs-NM) could be selectively quenched by Cu2+ ions in aqueous solution and consequently exhibited sensitive fluorescence “on–off” switching effects for Cu2+ ions. The detection limit was as low as 0.28 μM Cu2+, which was far below the U.S. EPA limit of 20 μM. This prefigured the potential applications of the current chemosensor for the detection of Cu2+ ions in aqueous solution and in living cells. The small-angle XRD, TEM, BET, FT-IR, UV-vis absorption measurements all agreed on the fact that the sensitive probes of BM had been successfully labelled at the pore surface of MSNs.

 Please wait while we load your content...
Please wait while we load your content...