Modeling intermediates in carbon monoxide coupling reactions using cyclooctatetraene thorium derivatives†
Abstract
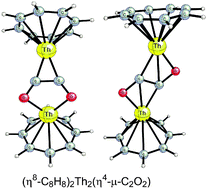
The interaction of carbon monoxide with organoactinides has recently been shown experimentally, particularly by Cloke and co-workers, to result in coupling to give the oligomeric anions CnOn2− (n = 2, 3, 4). In order to model possible intermediates in reactions of this type, we have used density functional theory to explore the systems (C8H8)Th(CO)n (n = 1 to 5) and (C8H8)2Th2(CO)n (n = 2 to 7) related to the known “thorocene”, (η8-C8H8)2Th. Thorium was chosen as the actinide for this work since its chemistry almost entirely involves the single diamagnetic +4 oxidation state. All of the binuclear (C8H8)2Th2(CO)n structures found in this work have long Th⋯Th distances ranging from 4.4 to 5.0 Å suggesting the absence of direct Th–Th bonds. Two (C8H8)2Th2(CO)2 isomers of similar energies in which the two CO groups have coupled to form trans and cis isomers of a bridging η4-μ-C2O2 ligand are low energy structures. These bridging η4-μ-C2O2 ligands exhibit ultralow ν(CO) frequencies around 1000 cm−1 indicating strong back donation of thorium d and f electrons into C–O antibonding orbitals. Most of the carbonyl richer (C8H8)2Th2(CO)n (n = 3 to 7) structures are derived from one of these basic (C8H8)2Th2(CO)2 structures by addition of terminal CO groups. An exception is the lowest energy (C8H8)2Th2(CO)4 structure which has C4v symmetry with four equivalent separate η2-μ-CO groups bridging the thorium atoms. The thermochemistry of these systems suggest (C8H8)Th(CO)4 and (C8H8)2Th2(CO)n (n = 2, 4) to be the most promising synthetic objectives, which are potentially obtainable by reductive carbonylation of the known (C8H8)ThCl2.

 Please wait while we load your content...
Please wait while we load your content...