Non-surfactant-assisted synthesis, size control and electrocatalytic activity of Cu nanoparticles immobilized on carbon spheres
Abstract
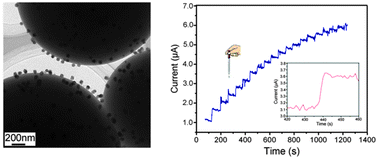
Carbon spheres (CSs) immobilized with monodispersed Cu nanoparticles were synthesized by a non-surfactant-assisted method. In this protocol, the biomass (ascorbic acid) transformed into the spheres of hydrothermal carbon through hydrothermal carbonization, and copper chlorite was reduced to Cu nanoparticles, which were in situ deposited on the spheres. No excess surfactant or capping reagent was necessary, which made the surface of the as-prepared nanoparticles very clean. In the following annealing, the spheres of hydrothermal carbon converted into CSs and the size of Cu nanoparticles could be tuned from several nanometers to dozens of nanometers by changing the annealing temperature only. This hybrid composite exhibited excellent catalytic activity for oxidation of glucose and could be employed as a rapid and inexpensive glucose sensor.

 Please wait while we load your content...
Please wait while we load your content...