Solid solvents: activation parameters for the rearrangement of cyclopropylcarbinyl bromide on mordenite zeolite
Abstract
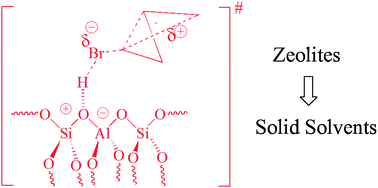
Rearrangement of cyclopropylcarbinyl bromide over proton and ammonium-exchanged mordenite (H-MOR and NH4-MOR) was studied at different temperatures. The product distribution analysis revealed a similar profile to what is found in solution, with prevalence of cyclobutyl over allylcarbinyl bromide. The activation parameters showed that compared with 80% aqueous ethanol solution, the zeolites present lower enthalpy of activation and higher entropy of activation, but yielding a significantly lower 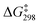 . The results may be explained in terms of the ability of zeolites to serve as solid solvents, providing a polar nanoenvironment for ionic reactions to occur.
. The results may be explained in terms of the ability of zeolites to serve as solid solvents, providing a polar nanoenvironment for ionic reactions to occur.

 Please wait while we load your content...
Please wait while we load your content...