A quinoxaline-fused tetrathiafulvalene derivative and its semiconducting charge-transfer salt: synthesis, crystal structures and physical properties†
Abstract
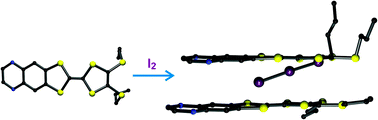
A quinoxaline-fused tetrathiafulvalene (TTF) derivative 1 has been synthesized to form a compact and planar π-conjugated donor–acceptor (D–π–A) ensemble, and its single crystal structure has been determined by X-ray diffraction. The inherent redox activity of 1 has been probed by cyclic voltammetry, and UV-vis spectroscopy revealed the typical broad and intense intramolecular charge-transfer (ICT) absorption occurring for such compactly fused D–π–A molecules. Reaction with iodine led to a 2 : 1 semiconducting charge-transfer salt {(1)2I3}, whose single crystal structure investigation, however, underlined the occurrence of a pronounced charge localization in the organic lattice. Consequently, the electrical conductivity of {(1)2I3}, measured by the four contact method on single crystals, gave only a limited value of about 1 × 10−4 Ω−1 cm−1, and the activation energy was determined to be on the order of 470–480 meV.
- This article is part of the themed collection: Advanced Complex Inorganic Nanomaterials

 Please wait while we load your content...
Please wait while we load your content...