Trivalent methylated arsenic metabolites induce apoptosis in human myeloid leukemic HL-60 cells through generation of reactive oxygen species
Abstract
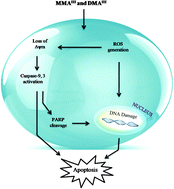
Arsenic trioxide (As2O3) has remarkable therapeutic efficacy against leukemia. However, after As2O3 biotransformation, the role of arsenic metabolites in the clinical efficacy against leukemia still needs to be elucidated. Therefore, to explore the contribution of trivalent methylated arsenicals in the therapeutic effects, we investigated and compared the effects of arsenite (iAsIII), monomethylarsonous acid (MMAIII) and dimethylarsinous acid (DMAIII) on HL-60 cells. Methylated arsenic species MMAIII and DMAIII showed potentially reduced cell survival with IC50 values of 3 and 2 μM, respectively. We found that methylated metabolites caused apoptosis through oxidative stress and loss of mitochondrial membrane potential. Furthermore, we found that the caspase-9 and -3 were markedly activated by exposure to methylated metabolites, with cleavage of poly-ADP ribose polymerase (PARP). Conversely, cellular apoptosis, generation of ROS, activation of caspase-3, -9 as well as PARP cleavage were significantly attenuated by pretreatment with an antioxidant, N-acetylcysteine (NAC). DNA damage was also markedly observed in HL-60 cells exposed to either MMAIII or DMAIII, while iAsIII did not show any relevant effects in HL-60 cells. Likewise, phosphorylation of the histone H2A variant (γ-H2AX), a biomarker of DNA damage, significantly occurred in cellular nuclei following exposure to two methylated species, which was reduced in the presence of NAC, suggesting that the induction of DNA damage was predominantly caused by the two metabolites via oxidative stress. In conclusion, we suggest that arsenic intermediate metabolites; MMAIII and DMAIII might prove to be of clinical relevance in future as such approaches may help in the treatment of leukemia and other types of cancers.

 Please wait while we load your content...
Please wait while we load your content...