Mechanism of cellular accumulation of an iridium(iii) pentamethylcyclopentadienyl anticancer complex containing a C,N-chelating ligand†
Abstract
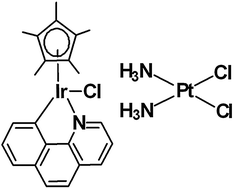
The effect of replacement of the N,N-chelating ligand 1,10-phenanthroline (phen) in the IrIII pentamethylcyclopentadienyl (Cp*) complex [(η5-Cp*)(Ir)(phen)Cl]+ (2) with the C,N-chelating ligand 7,8-benzoquinoline (bq) to give [(η5-Cp*)(Ir)(bq)Cl] (1) on the cytotoxicity of these Cp*IrIII complexes toward cancer cell lines was investigated. Complex 2 is inactive, similar to other Cp*IrIII complexes containing the N,N-chelating ligands. In contrast, a single atom change (C− for N) in the chelating N,N ligand resulted in potency in human ovarian carcinoma cisplatin-sensitive A2780 cells, and, strikingly, 1 is active in the cisplatin-resistant human breast cancer MCF-7 and A2780/cisR cells. Replacement of the N,N-chelating ligand with the C,N-chelating ligand gives rise to increased hydrophobicity, leading to higher cellular accumulation, higher DNA-bound iridium in cells and higher cytotoxicity. The pathways involved in cellular accumulation of 1 have been further explored and compared with conventional cisplatin. The results show that both energy-independent passive diffusion and energy-dependent transport play a role in accumulation of 1. Further results were consistent with involvement of p-glycoprotein, multidrug resistance-associated protein 1 and glutathione metabolism in the efflux of 1. In contrast, the internalization of 1 mediated by the endocytotic uptake pathway(s) seems less likely. Understanding the factors which contribute to the mechanism of cellular accumulation of this IrIII complex can now lead to the design of structurally similar metal complexes for antitumor chemotherapy.

 Please wait while we load your content...
Please wait while we load your content...