Multivalent amino sugars to recognize different TAR RNA conformations†
Abstract
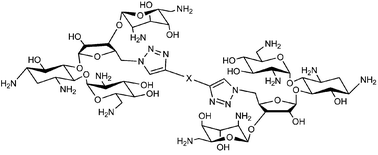
Neomycin dimers synthesized using “click chemistry” with varying functionality and length in the linker region have been shown to be effective in targeting the HIV-1 transactivation response element (TAR) RNA region of the HIV virus. TAR, a 59 base pair stem loop structure located at the 5′-end of all nascent viral transcripts interacts with its target, a key regulatory protein, Tat, and necessitates the replication of HIV-1 virus. Ethidium bromide displacement and FRET competition assays have revealed nanomolar binding affinity between neomycin dimers and wildtype TAR RNA while in case of neomycin, only a weak binding was detected. Here, NMR and FID-based comparisons reveal an extended binding interface for neomycin dimers involving the upper stem of the TAR RNA thereby offering an explanation for increased affinities. To further explore the potential of these modified aminosugars we have extended binding studies to include four TAR RNA mutants that display conformational differences with minimal sequence variation. The differences in binding between neomycin and neomycin dimers is characterized with TAR RNA mutants that include mutations to the bulge region, hairpin region, and both the bulge and hairpin regions. Our results demonstrate the effect of these mutations on neomycin binding and our results show that linker functionalities between dimeric units of neomycin can distinguish between the conformational differences of mutant TAR RNA structures.

 Please wait while we load your content...
Please wait while we load your content...