Conformational properties of cholic acid, a lead compound at the crossroads of bile acid inspired drug discovery†
Abstract
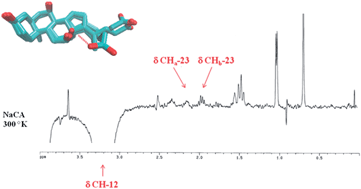
Cholic acid is an endogenous primary bile acid endowed with endocrine signaling functions and digestive properties. In this work its conformational profile and the impact of the steroid nucleus are investigated using DFT studies and NMR spectroscopy. As a result, three major minima conformations are found with ‘extended’, ‘semi-extended’ and ‘folded’ side chains, respectively. While the ‘extended’ and ‘semi-extended’ conformations are often found as bioactive conformations in the binding site of co-crystallized biological targets, the ‘folded’ conformation is only experimentally observed in NMR studies. Overall, the results of this study provide additional basis for the interpretation of biological functions of bile acids, and disclose new pharmacophoric templates for the design of constrained bile acid analogues.

 Please wait while we load your content...
Please wait while we load your content...