A novel approach to high pressure flow digestion
Abstract
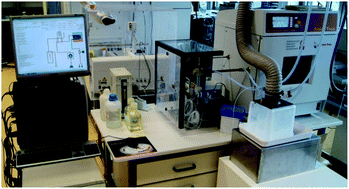
A new high pressure flow digestion system has been developed for sample digestion at a pressure of up to 40 bar and a temperature of about 230 °C. The reaction with acids takes place in a PFA tube and is heated by microwave radiation in a multimode cavity. As the PFA tube cannot withstand the harsh digestion conditions without support, it is placed inside a coiled glass tube pressurized by 40 bar nitrogen thus forming an autoclave. Corrosion of system components by acid fumes and related sample contamination is circumvented by establishing a slow but steady flow of the high pressure nitrogen countercurrent to the sample flow. The presented system does not constrain the selection of the digestion reagent. Acid cocktails of nitric acid with hydrochloric and/or hydrofluoric acid as well as hydrogen peroxide were successfully used for the digestion of various samples. The method accuracy was validated with five certified reference materials (BCR 62, DORM-2, NIST SRM 1515, NIST SRM 1567, NIST SRM 1568) and good agreement between the determined and the certified values was obtained for Al, Ca, Cr, Cu, Fe, Mg, Mn, Ni, Pb, and Zn using inductively coupled plasma optical emission spectrometry (ICP-OES) for analyte quantification. The flow digestion of the CRMs resulted in clear solutions with residual carbon concentrations (RCC) between 11 and 40%. Spike recoveries of Al, As, Ba, Be, Bi, Cd, Co, Cr, Cu, Fe, Mg, Mn, Mo, Ni, Pb, Sb, Se, Sr, Ti, V, and Zn were between 94 and 105%. For Hg the spike recovery was 89%. The fully automated high pressure flow digestion system is capable of digesting up to 6 samples per hour.
- This article is part of the themed collection: 2014 Young Analytical Scientists

 Please wait while we load your content...
Please wait while we load your content...