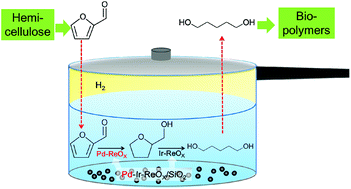
One-pot selective conversion of furfural into 1,5-pentanediol (1,5-PeD) was carried out over Pd-added Ir–ReOx/SiO2catalysts through two-step reaction temperatures. The Pd(0.66 wt%)–Ir–ReOx/SiO2catalyst showed the best performance in the production of 1,5-PeD from furfural. The maximum yield of 1,5-PeD was 71.4%. The furfural conversion and yield of 1,5-PeD was almost maintained during four repeated tests when the catalyst was calcined again. The characterization results from TPR, XRD, XANES, EXAFS and FT-IR of adsorbed CO indicated that Pd–Ir–ReOx/SiO2catalysts consisted of ReOx-modified Pd metal particles and ReOx-modified Ir metal particles. The lower-temperature reaction step was very crucial for the total hydrogenation of furfural into a tetrahydrofurfuryl alcohol intermediate, which was converted into 1,5-PeD by hydrogenolysis during the high temperature step over the ReOx-modified Ir metal particles.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...