Hydrogen tungsten bronze as a decoking agent for long-life, natural gas-fueled solid oxide fuel cells†
Abstract
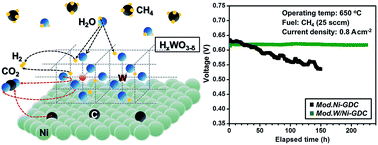
Ni catalyst-based anodes in solid oxide fuel cells (SOFCs) operating with natural gas fuel are vulnerable to coking (carbon deposition) and sulfur poisoning, resulting in impaired anodes and eventual cessation of SOFC operation. The incorporation of as small as 1 atom% W to Ni is found to overcome this formidable challenge. The hydrogen tungsten bronze (HxWO3−δ ≡ WO3−x−δ(OH)x) formed by reaction with the water vapor produced during SOFC operation supplies the hydroxyl groups to oxidize carbon and eliminate coking on the Ni surface. Mobile protons adhered to the oxide ions as well as the mobile oxide ions themselves in the AO3 sublattice of the oxygen-deficient perovskite-type HxWO3−δ facilitate a rapid cleaning of the Ni surface. We show here that Ni–HxWO3−δ–Ce0.8Gd0.2O1.9 (GDC) anode-supported single cell SOFCs with GDC electrolyte and Ba0.5Sr0.5Co0.8Fe0.2O3−δ cathode exhibit more than 200 h of stable operation with methane fuel at 650–700 °C.

 Please wait while we load your content...
Please wait while we load your content...