Non-nitrogen doped and non-metal oxygen reduction electrocatalysts based on carbon nanotubes: mechanism and origin of ORR activity†
Abstract
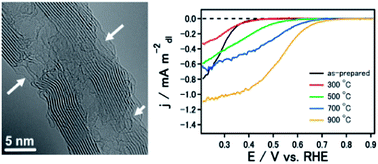
We show high activity in non-nitrogen doped and non-metal electrocatalysts based on carbon nanotubes with onset potential up to 0.73 V vs. RHE by the formation of hole defects on the walls of carbon nanotubes, followed by annealing under Ar atmosphere. From the power generation test, this catalyst can deliver a maximum output power of 55.68 mW (mg−1CNT cathode). Through temperature programmed desorption (TPD) and electrochemical analysis, the creation of new active sites is correlated with the removal of high temperature CO desorbing functionalities. Residual metal impurities were examined by the use of inductively coupled plasma-mass spectroscopy (ICP-MS), high angle annular dark-field (HAADF) and electron energy loss (EELS) analysis. The extremely low amount of metal impurities and the absence of impurity coordination at the edge planes after electrochemical characterization suggest that it is unlikely that impurities directly contribute to ORR. We conclude by proposing that the origin of ORR activity is a result of carbon restructuring and the possible formation of topological defects during the removal of high temperature CO desorbing functional groups.

 Please wait while we load your content...
Please wait while we load your content...