New insights into the chemistry of di- and trimetallic iron dithiolene derivatives. Structural, Mössbauer, magnetic, electrochemical and theoretical studies†‡
Abstract
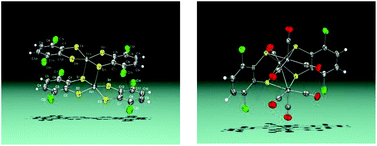
Reaction of Fe3(CO)12 with 1,2-dithiolene HSC6H2Cl2SH affords a mixture of complexes [Fe2(CO)6(μ-SC6H2Cl2S)] 1, [Fe2(SC6H2Cl2S)4] 2 and [Fe3(CO)7(μ3-SC6H2Cl2S)2] 3. In the course of the reaction the trimetallic cluster 3 is first formed and then converted into the known dinuclear compound 1 to afford finally the neutral diiron tetrakis(dithiolato) derivative 2. Compounds 2 and 3 have been studied by Mössbauer spectroscopy, X-ray crystallography and theoretical calculations. In compound 2 the metal atoms are in an intermediate-spin FeIII state (SFe = 3/2) and each metal is bonded to a bridging dithiolene ligand and a non-bridging thienyl radical (S = 1/2). Magnetic measurements show a strong antiferromagnetic coupling in complex 2. Cyclic voltammetry experiments show that the mixed valence trinuclear cluster 3 undergoes a fully reversible one electron reduction. Additionally, compound 3 behaves as an electrocatalyst in the reduction process of protons to hydrogen.

 Please wait while we load your content...
Please wait while we load your content...