Bismuth(iii) complexes derived from α-amino acids: the impact of hydrolysis and oxido-cluster formation on their activity against Helicobacter pylori†
Abstract
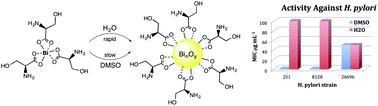
Eight bismuth(III) complexes derived from a variety of α-amino acids covering a range of physico-chemical properties (L-phenylalanine (Phe), L-proline (Pro), L-methionine (Met), L-cysteine (Cys), D,L-serine (Ser), L-tyrosine (Tyr), L-aspartic acid (Asp) and L-glutamic acid (Glu)) have been synthesised, characterised, and evaluated for their activity against Helicobacter pylori. The optimal synthetic procedure utilises [Bi(OtBu)3], giving the complexes [BiL3] (L = Phe 1, Pro 2, Met 3, Ser 5, Tyr 6) and [Bi2L3] (L = Cys 4, Asp 7, Glu 8) cleanly and in good yield. However, the synthesis is sensitive to both temperature and moisture. The solubility and stability of the bismuth(III) complexes was investigated using ESI-MS. Almost all compounds (except for [Bi(Phe)3] and [Bi(Pro)3]) were found to be partially or completely soluble in aqueous solution giving a pH 2.5–5.0, indicating the presence of free α-amino acid and hydrolysis of the bismuth(III) complexes to polynuclear bismuth oxido-clusters. The results of the bactericidal studies against Helicobacter pylori demonstrate that this hydrolysis process impacts significantly on the observed Minimum Inhibitory Concentration (MICs) which are increased substantially, often by many orders of magnitude, when the complexes are initially prepared in water rather than DMSO.

 Please wait while we load your content...
Please wait while we load your content...