C–H activation and metalation at electrode surfaces: 2,3-dimethyl-1,4-dihydroxybenzene on Pd(pc) and Pd(111) studied by TLE, HREELS and DFT
Abstract
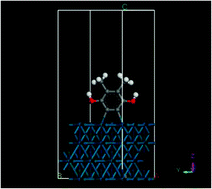
Previous studies, based on thin-layer electrochemistry (TLE), in situ scanning tunneling microscopy (EC-STM), high-resolution electron energy loss spectroscopy (HREELS) and density functional theory (DFT) computations, on the chemical adsorption of hydroquinone from aqueous solutions onto atomically smooth Pd (and Pt) electrode surfaces indicated two modes of attachment that depended upon the solution concentration. At low activities, the diphenol was oxidatively chemisorbed as benzoquinone in a flat orientation, suggestive of a Pd(2,3,5,6-η-C6H4O2) surface complex; at higher concentrations, vertical chemisorption was effected via two C–H bond activations (or metalations) at the 2 and 3 ring positions, evocative of an o-phenylene organopalladium compound. We have extended the work to 2,3-dimethyl-1,4-dihydroxybenzene on Pd(pc) and Pd(111) electrodes to probe the effect of two methyl substituents on only one side of the diphenol ring. Surface coverage and adsorbed-molecule cross section data from TLE and HREELS measurements revealed non-random concentration-dependent adsorbate orientations similar to the oxidative chemisorption of hydroquinone: flat at low concentrations and edgewise at elevated concentrations. The DFT results suggested that, for the flat structure, surface coordination is via the two double bonds of the quinone ring as in [Pd(2,3,5,6-η)-2,3-dimethyl-p-quinone]. For the edge-vertical orientation, a structure analogous to an o-phenylene compound is generated in which C–H bonds at the 5 and 6 ring positions are activated and then metalated. DFT-simulated HREELS spectra helped identify the observed peaks that distinguish the surface-coordinated quinone from the surface-metalated diphenol.
- This article is part of the themed collection: The Carbon–Metal Bond and C–H Metalation

 Please wait while we load your content...
Please wait while we load your content...