Aggregation-induced white-light emission from the triple-stranded dinuclear Sm(iii) complex†
Abstract
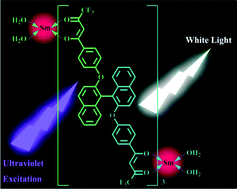
A novel bis-β-diketone ligand, 4,4′-bis(4,4,4-trifluoro-1,3-dioxobutyl)(phenoxy)-1,1′-binaphthalene (BTPB), is designed for synthesis of a white light emissive lanthanide complex. The ligand bears two benzoyl β-diketonate sites linked by a 1,1′-binaphthoxy spacer. Reaction of the doubly negatively charged bis-bidentate ligand with lanthanide ions forms triple-stranded dinuclear complexes Sm2(BTPB)3(H2O)4 (1) and Gd2(BTPB)3(H2O)4 (2), which have been fully characterized by various spectroscopic techniques. UV-Vis absorption and emission spectroscopic techniques are used to investigate photophysical properties of the ligand and its complexes in THF and CHCl3. In some cases aggregation of the ligand results in the appearance of a new luminescence band at about 510 nm in addition to the monomer fluorescence. In complex 1, partial energy transfer from BTPB results in Sm(III)-based red light emission in addition to the BTPB-based blue/green emission. With the variation of the excited wavelength and concentration of the solution, complex 1 shows a tunable white light emission with the balance of three primary colors. This is an unusual case of observation of white light emission from a single molecule Sm(III) complex.

 Please wait while we load your content...
Please wait while we load your content...