Mechanistic studies of CO2 reduction to methanol mediated by an N-heterocyclic germylene hydride†
Abstract
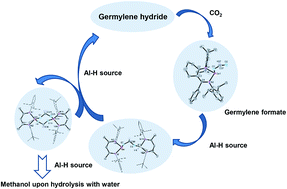
The labile germylene hydride LCyGeH is capable of activating CO2 affording the corresponding formate LCyGeOCH(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) (2) (LCy = cyclo-C6H8-1-NAr-2-C(Ph)NAr, Ar = 2,6-iPr2C6H3). Compound 2 and the previously reported LGeOCH(
O) (2) (LCy = cyclo-C6H8-1-NAr-2-C(Ph)NAr, Ar = 2,6-iPr2C6H3). Compound 2 and the previously reported LGeOCH(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) (L = CH(MeC
O) (L = CH(MeC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NAr)2, Ar = 2,6-iPr2C6H3) (2′) could be further converted to methanol with the AlH3·NMe3 alane-amine adduct as a hydrogen source upon workup with water. A plausible mechanism for the conversion of the formate complexes to methanol is proposed based on additional results from the conversion of 2′ with the milder hydride delivery agent LAlH2.
NAr)2, Ar = 2,6-iPr2C6H3) (2′) could be further converted to methanol with the AlH3·NMe3 alane-amine adduct as a hydrogen source upon workup with water. A plausible mechanism for the conversion of the formate complexes to methanol is proposed based on additional results from the conversion of 2′ with the milder hydride delivery agent LAlH2.

 Please wait while we load your content...
Please wait while we load your content...