Synthesis and characterisation of new MO(OH)2 (M = Zr, Hf) oxyhydroxides and related Li2MO3 salts
Abstract
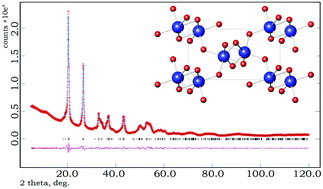
Two new solid MO(OH)2 (M = Zr, Hf) oxyhydroxides have been synthesised by an ion-exchange reaction from Li2MO3 (M = Zr, Hf) precursors obtained by a citrate combustion technique. The crystal structure of the oxyhydroxides has been solved by direct methods and refined using Rietveld full profile fitting based on X-ray powder diffraction data. Both oxyhydroxides crystallize in a P21/c monoclinic unit cell and have a structure resembling that of the related salts. Detailed characterisation of the fine-structure features and chemical bonding in precursors and oxyhydroxide powders has been performed using vibrational spectroscopy, nuclear magnetic resonance spectroscopy, scanning electron microscopy, pair distribution function analysis and quantum-chemical modelling.

 Please wait while we load your content...
Please wait while we load your content...