p-Tolylimido rhenium(v) complexes – synthesis, X-ray studies, spectroscopic characterization, DFT calculations and catalytic activity†
Abstract
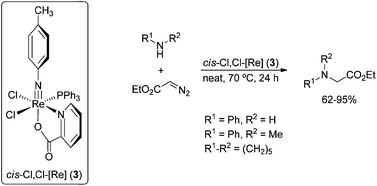
Novel p-tolylimido rhenium(V) complexes trans-(Cl,Cl)-[Re(p-NC6H4CH3)Cl2(py-2-COO)(PPh3)]·MeCN (1a), trans-(Cl,Cl)-[Re(p-NC6H4CH3)Cl2(py-2-COO)(PPh3)] (1b), trans-(Br,Br)-[Re(p-NC6H4CH3)Br2(py-2-COO)(PPh3)] (2), cis-(Cl,Cl)-[Re(p-NC6H4CH3)Cl2(py-2-COO)(PPh3)] (3), cis-(Br,Br)-[Re(p-NC6H4CH3)Br2(py-2-COO)(PPh3)]·H2O (4), trans-(Cl,Cl)-[Re(p-NC6H4CH3)Cl2(OMe)] (5) and trans-(Br,Br)-[Re(p-NC6H4CH3)Br2(OMe)(PPh3)2]·1/2H2O (6) were synthesized. The compounds were identified by elemental analysis, IR, NMR (1H, 13C and 31P), UV-Vis spectroscopy and X-ray crystallography. Calculations at the DFT level were undertaken in order to elucidate the structural, spectroscopic and bonding properties of the trans-(Cl,Cl) and cis-(Cl,Cl) isomers of [Re(p-NC6H4CH3)Cl2(py-2-COO)(PPh3)]. In the first step, extended tests of the ability of different DFT methods (B3LYP, BP86, and PBE1PBE) were performed to find the best overall performer in describing the geometry of the imido Re(V) complexes; then, the electronic spectra of the trans and cis isomers of [Re(p-NC6H4CH3)Cl2(py-2-COO)(PPh3)] were investigated at the TDDFT level employing the PBE1PBE functional. Additional information about bonding in the compounds [Re(p-NC6H4CH3)Cl2(py-2-COO)(PPh3)] was obtained by NBO analysis. The catalytic activity of the complexes incorporating the picolinate ligand was studied and compared in the synthesis of N-substituted ethyl glycine esters from ethyl diazoacetate and amines.

 Please wait while we load your content...
Please wait while we load your content...