Cyclometalated heteronuclear Pt/Ag and Pt/Tl complexes: a structural and photophysical study†
Abstract
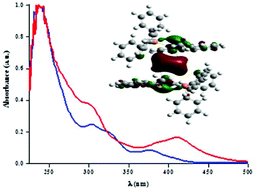
To investigate the factors influencing the luminescent properties of polymetallic cycloplatinated complexes a detailed study of the photophysical and structural properties of the heteronuclear complexes [Pt2Me2(bhq)2(μ-dppy)2Ag2(μ-acetone)](BF4)2, 2, [PtMe(bhq)(dppy)Tl]PF6, 3, and [Pt2Me2(bhq)2(dppy)2Tl]PF6, 4, [bhq = benzo[h]quinoline, dppy = 2-(diphenylphosphino)pyridine] was conducted. Complexes 3 and 4 synthesized by the reaction of [PtMe(bhq)(dppy)], 1, with TlPF6 (1 or 1/2 equiv.) and stabilized by unsupported Pt–Tl bonds as revealed by multinuclear NMR spectroscopy and confirmed by X-ray crystallography for 3. DFT calculations for the previously reported butterfly Pt2Ag2 cluster 2 reveal that in the optimized geometry the bridging acetone molecule is removed and the metal core displays a planar-shaped geometry in which according to a QTAIM calculation and natural bond orbital (NBO) analysis the Ag⋯Ag metallophilic interaction is strengthened. In contrast to the precursor 1, which is only emissive in glassy solutions (3MLCT 485 nm), all 2–4 heteropolynuclear complexes display intense emissions in the solid state and in glassy solutions. Time-dependent density functional theory (TD-DFT) is used to elucidate the origin of the electronic transitions in the heteronuclear complexes 2 and 3. The low energy absorption and intense orange emission for cluster 2 (solid 77 K and glass) are attributed to metal–metal to ligand charge transfer (MM′LCT) with a minor L′LCT contribution. For 3 and 4 two different bands are developed: the high energy band (602–630 nm) observed for 4 at 77 K (solid, glass) and in diluted glasses for 3 is ascribed to emission from discrete Pt2Tl units of mixed 3L′LCT/3LM′CT origin. However, the low energy band (670–690 nm) observed at room temperature (solid) for both complexes and also in concentrated glasses for 3 is assigned to 3ππ excited states arising from intermolecular interactions.

 Please wait while we load your content...
Please wait while we load your content...