Nucleophile-directed selectivity towards linear carbonates in the niobium pentaethoxide-catalysed cycloaddition of CO2 and propylene oxide†
Abstract
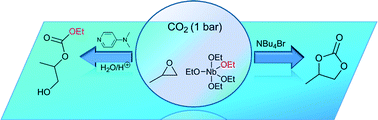
Homoleptic Nb-complexes combined with selected organic nucleophiles generate very active catalytic systems for the cycloaddition of propylene oxide and CO2 under ambient conditions. An unprecedented reaction pathway towards an acyclic organic carbonate is observed when extending the study to [Nb(OEt)5] in combination with 4-dimethylamino-pyridine (DMAP) or tetra-n-butylammonium bromide (TBAB). Mechanistic insights of the reaction are provided based on experimental and spectroscopic evidences.
- This article is part of the themed collection: Carbon dioxide conversion

 Please wait while we load your content...
Please wait while we load your content...