Na2HPO4-modified NaY nanocrystallites: efficient catalyst for acrylic acid production through lactic acid dehydration†
Abstract
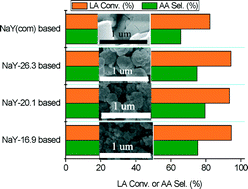
By regulating the H2O/SiO2 molar ratio (n = 13.8, 16.9, 20.1, 26.3), NaY zeolites of different particle sizes (NaY-n, 50–400 nm) were synthesized, and NaY-n was modified with disodium hydrogen phosphate, Na2HPO4. The as-prepared catalysts are highly effective for the dehydration of sustainable lactic acid (LA) to produce acrylic acid (AA). Through the optimization of NaY particle size and Na2HPO4 loading, AA yields as high as 74.3% can be achieved under mild conditions (340 °C). In terms of employed feed rate of lactic acid and amount of catalyst, the AA formation rate is 12.0 mmol gcat−1 h−1, the highest ever reported for LA dehydration to AA. Employing slightly low Si/Al ratios in NaY-n can modify catalyst acidity, while pre-applied Na+ ion-exchange and following Na2HPO4 loading can effectively reduce the number of acid sites, particularly the Brønsted ones on NaY-n. Equally important structural features of NaY-n are their shorter pore channels (or even incompletely developed zeolite texture) and relatively larger Sexternal/SBET ratio, which favors quick product departure from the catalyst surface, and reduces the possibility of side reactions occurring inside the long channels. The appropriate surface acidity together with unique structural features of NaY-n ensure efficient LA conversion with high dehydration selectivity. Higher surface concentrations of Na+ on Na2HPO4/NaY-n also favor sodium lactate meanwhile suppressing poly-lactate as well as preventing carbon formation, corresponding to superior performance as well as improved durability of Na2HPO4/NaY-n.

 Please wait while we load your content...
Please wait while we load your content...