On the near UV photophysics of a phenylalanine residue: conformation-dependent ππ* state deactivation revealed by laser spectroscopy of isolated neutral dipeptides†
Abstract
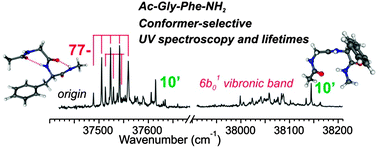
The primary step of the near UV photophysics of a phenylalanine residue is investigated in one- and two-color pump–probe R2PI nanosecond experiments carried out on specific conformers of the Ac-Gly-Phe-NH2 molecule and related neutral compounds isolated in a supersonic expansion. Compared to toluene, whose ππ* state photophysics is dominated by intersystem crossing with a lifetime of ∼80 ns at the origin, the first ππ* state of Phe in the peptide environment is systematically found to be shorter-lived. The lifetime at the origin of transition is found to be significantly shortened in the presence of a primary amide (–CONH2) group (20–60 ns, depending on the conformer considered), demonstrating the existence of an additional non-radiative relaxation channel related to this chemical group. The quenching effect induced by the peptide environment is still more remarkable beyond the origin of the ππ* state, since vibronic bands of one of the 4 conformers observed (the 27-ribbon conformation) become barely detectable in the ns R2PI experiment, suggesting a significant conformer-selective lifetime shortening (below 100 ps). These results on dipeptides, which extend previous investigations on shorter Phe-containing molecules (N-Ac-Phe-NH2 and N-Ac-Phe-NH-Me), confirm the existence of conformer-dependent non-radiative deactivation processes, whose characteristic timescales range from tens of ns down to hundreds of ps or below. This dynamics is assigned to two distinct mechanisms: a first one, consistent with an excitation energy transfer from the optically active ππ* state to low-lying amide nπ* excited states accessed through conical intersections, especially in the presence of a C-terminal primary amide group (–CONH2); a second one, responsible for the short lifetimes in 27 ribbon structures, would be more specifically triggered by phenyl ring vibrational excitations. Implications in terms of spectroscopic probing of Phe in a peptide environment, especially in the presence of a quenching amide group, are discussed.

 Please wait while we load your content...
Please wait while we load your content...