Destructuring ionic liquids in ionogels: enhanced fragility for solid devices
Abstract
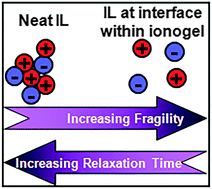
Confining ionic liquids (ILs) with added lithium salt within silica host networks enhances their fragility and improves their conductivity. Overall, conductivity measurements, Raman spectroscopy of the TFSI anion and NMR spectroscopy of the lithium cation show segregative interaction of lithium ions with the SiO2 host matrix. This implies at IL/SiO2 interfaces a breakdown of aggregated regions that are found systematically in bulk ILs. Such destructuration due to the interface effect determines the fragility and thus results locally at the interface in short relaxation times, low viscosity, and good ionic conductivity. The “destructuration” of ion pairs or domains makes ILs within ionogels a competitive alternative to existing solid ionic conductors in all-solid devices, such as lithium batteries and supercapacitors.

 Please wait while we load your content...
Please wait while we load your content...