Towards the ionic limit of two-dimensional materials: monolayer alkaline earth and transition metal halides†
Abstract
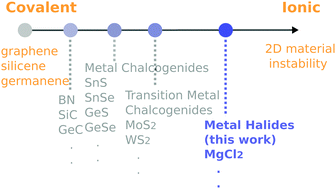
We theoretically explored new two-dimensional materials near the ionic instability (three-dimensional structures are favored), with covalent bonded systems (graphene) sitting at the opposite end of the spectrum. Accordingly, monolayer alkaline earth and transition metal halides, many of their bulk forms being layered structures, were investigated by density functional calculations. We thus predicted a new class of two-dimensional materials by performing structure relaxation, cohesive/formation energy and full phonon dispersion calculations. These materials exhibit strong ionic bonding character, as revealed by significant charge transfers. The superior charge donating/accepting abilities and the large specific area make these new materials promising for adsorption and catalytic reactions. We demonstrated adsorption and diffusion of Li on these materials, which are relevant for Li ion battery electrodes and hydrogen storage. Also the new materials with varied charge donating abilities and their nanostructures can enhance and tune catalytic reactions, such as Ziegler–Natta catalysts. Moreover, they exhibit diverse electronic properties that can be of great application interest, ranging from insulators to metals, and even spin-polarized semiconductors.

 Please wait while we load your content...
Please wait while we load your content...