Electrical conductivity and glass formation in nitrile-functionalized pyrrolidinium bis(trifluoromethylsulfonyl)imide ionic liquids: chain length and odd–even effects of the alkyl spacer between the pyrrolidinium ring and the nitrile group
Abstract
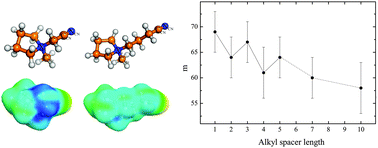
The electrical conductivity of a series of pyrrolidinium bis(trifluoromethylsulfonyl)imide ionic liquids, functionalized with a nitrile (cyano) group at the end of an alkyl chain attached to the cation, was studied in the temperature range between 173 K and 393 K. The glass formation of the ionic liquids is influenced by the length of the alkyl spacer separating the nitrile function from the pyrrolidinium ring. The electrical conductivity and the viscosity do not show a monotonic dependence on the alkyl spacer length, but rather an odd–even effect. An explanation for this behavior is given, including the potential energy landscape picture for the glass transition.

 Please wait while we load your content...
Please wait while we load your content...