Kinetics study of heterogeneous reactions of ozone with erucic acid using an ATR-IR flow reactor
Abstract
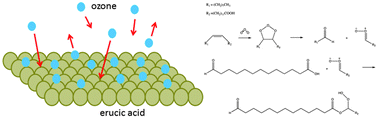
The ozone initiated heterogeneous oxidation of erucic acid (EA) thin film was investigated using a flow system combined with attenuated total reflection infrared spectroscopy (ATR-IR) over wide ranges of ozone concentrations (0.25–60 ppm), thin film thickness (0.1–1.0 μm), temperatures (263–298 K), and relative humidities (0–80% RH) for the first time. Pseudo-first-order rate constants, kapp, and overall reactive uptake coefficients, γ, were obtained through changes in the absorbance of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching bands at 1695 cm−1, which is assigned to the carbonyl group in carboxylic acid. Results showed that the reaction followed the Langmuir–Hinshelwood mechanism and kapp was largely dominated by surface reaction over bulk phase reaction. In addition, both the kapp and the γ values showed very strong temperature dependences (∼two orders of magnitude) over the temperature range; in contrast, they only slightly increased with increasing RH values from 0–80%. According to the kapp values as a function of temperature, the activation energy for the heterogeneous reaction was estimated to be 80.6 kJ mol−1. Our results have suggested that heterogeneous reactions between ozone and unsaturated solid surfaces likely have a substantially greater temperature dependence than liquid ones. Moreover, the hygroscopic properties of EA thin films before and after exposure to ozone were also studied by measurement of water uptake. Based on the hygroscopicity data, the insignificant RH effect on reaction kinetics was probably due to the relatively weak water uptake by the unreacted and reacted EA thin films.
O stretching bands at 1695 cm−1, which is assigned to the carbonyl group in carboxylic acid. Results showed that the reaction followed the Langmuir–Hinshelwood mechanism and kapp was largely dominated by surface reaction over bulk phase reaction. In addition, both the kapp and the γ values showed very strong temperature dependences (∼two orders of magnitude) over the temperature range; in contrast, they only slightly increased with increasing RH values from 0–80%. According to the kapp values as a function of temperature, the activation energy for the heterogeneous reaction was estimated to be 80.6 kJ mol−1. Our results have suggested that heterogeneous reactions between ozone and unsaturated solid surfaces likely have a substantially greater temperature dependence than liquid ones. Moreover, the hygroscopic properties of EA thin films before and after exposure to ozone were also studied by measurement of water uptake. Based on the hygroscopicity data, the insignificant RH effect on reaction kinetics was probably due to the relatively weak water uptake by the unreacted and reacted EA thin films.

 Please wait while we load your content...
Please wait while we load your content...