Chemical composition tuning in quaternary p-type Pb-chalcogenides – a promising strategy for enhanced thermoelectric performance†
Abstract
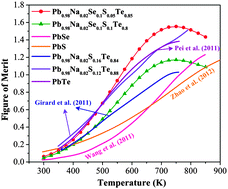
Recently a significant improvement in the thermoelectric performance of p-type ternary PbTe–PbSe and PbTe–PbS systems has been realized through alternating the electronic band structure and introducing nano-scale precipitates to bulk materials respectively. However, the quaternary system of PbTe–PbSe–PbS has received less attention. In the current work, we have excluded phase complexity by fabricating single phase sodium doped PbTe, alloyed with PbS up to its solubility limit which is extended to larger concentrations than in the ternary system of PbTe–PbS due to the presence of PbSe. We have presented a thermoelectric efficiency of approximately 1.6 which is superior to ternary PbTe–PbSe and PbTe–PbS at similar carrier concentrations and the binary PbTe, PbSe and PbS alloys. The quaternary system shows a larger Seebeck coefficient than the ternary PbTe–PbSe alloy, indicative of a wider band gap, valence band energy offset and heavier carriers effective mass. In addition, the existence of PbS in the alloy further reduces the lattice thermal conductivity originated from phonon scattering on solute atoms with high contrast atomic mass. Single phase quaternary PbTe–PbSe–PbS alloys are promising thermoelectric materials that provide high performance through adjusting the electronic band structure by regulating chemical composition.

 Please wait while we load your content...
Please wait while we load your content...