On the lithiation reaction of niobium oxide: structural and electronic properties of Li1.714Nb2O5
Abstract
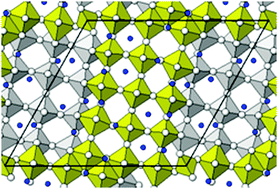
Monoclinic α-Nb2O5 was chemically lithiated by reaction with n-butyllithium, mimicking the product of electrochemical discharge of a niobium oxide cathode vs. a Li anode. The compound was investigated by neutron powder diffraction (D2B equipment at ILL, France) and its structure was Rietveld refined in space group P2 to wRp = 0.045, locating the Li atoms inserted in the α-Nb2O5 framework. The ensuing chemical formula is Li12/7Nb2O5. Some Li atoms are more strongly bonded (five coordinated O atoms), some are less strongly bonded (coordination number = 4). Starting from the experimental structure, first-principles periodic DFT calculations based on the hybrid B3LYP functional were performed. The electrochemical voltage of Li insertion was computed to be 1.67 V, fully consistent with the experimental 1.60 V plateau vs. capacity. The analysis of the electron band structure shows that lithiation changes the insulating oxide into a semi-metal; some of the extra electrons inserted with lithium become spin-polarized and give the material weak ferromagnetic properties.

 Please wait while we load your content...
Please wait while we load your content...