Mode-specific vibrational relaxation of photoexcited guanosine 5′-monophosphate and its acid form: a femtosecond broadband mid-IR transient absorption and theoretical study†
Abstract
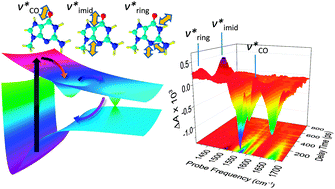
UV-pump/broadband-mid-IR-probe transient absorption (TA) experiments and ab initio quantum mechanical (QM) calculations were used to investigate the photophysics in heavy water of the neutral and acid forms of guanosine 5′-monophosphate (GMP and GMPD+, respectively). Excited GMP undergoes ultrafast internal conversion (IC) and returns to the electronic ground state in less than one picosecond with a large amount of excess vibrational energy. The spectroscopic signals are dominated by vibrational cooling – a process in which the solute dissipates vibrational energy to the solvent. For neutral GMP, cooling proceeds with a time constant of 3 ps. Following IC, at least some medium-frequency modes such as the carbonyl stretch and an in-plane ring vibration are excited, suggesting that the vibrational energy distribution is non-statistical. This is consistent with predicted structural changes upon passage through the S1/S0 conical intersection. GMPD+ differs from GMP by a single deuteron at the N7 position, but has a dramatically longer lifetime of 200 ps. Vibrational cooling of the S1 state of GMPD+ was monitored via several medium-frequency modes that were assigned using QM calculations. These medium-frequency modes are also vibrationally excited in a non-statistical fashion. Excitation of these modes is in line with the change in geometry at the S1 minimum of GMPD+ predicted by QM calculations. Furthermore, these modes relax at different rates, fully consistent with QM calculations, which predict that excited vibrational states of the carbonyl stretch couple strongly to the D2O solvent and thus deactivate via intermolecular energy transfer (IET). In contrast, the ring stretch couples strongly to other ring modes of the guanine chromophore and appears to decay via intramolecular vibrational energy redistribution (IVR).

 Please wait while we load your content...
Please wait while we load your content...