Insight into the general rule for the activation of the X–H bonds (X = C, N, O, S) induced by chemisorbed oxygen atoms†
Abstract
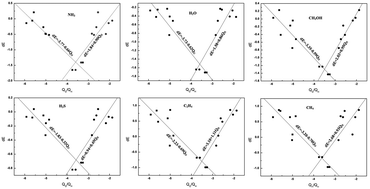
Density functional theory calculations are presented for adsorption and dissociation of NH3, H2O, CH3OH, H2S and C2H4 on clean and oxygen atom pre-adsorbed metal surfaces (Cu, Ag, Au, Ni, Pd, Pt, Rh, Ru, Os and Ir). The calculation results indicated that the oxygen-promotion effect depends both on the metallic activity and the character of the X–H bond. On the one hand, for a given reaction on a series metals, a good linear correlation was found between the energy barrier difference of X–H bond breaking on clean and oxygen-covered metals and the binding strength of oxygen on metals, namely an oxygen-promotion effect was favorable to the less active metals but unfavorable to the more active metals. On the other hand, for a series of X–H bond breaking reactions on a given metal, it was found that the promotion effect follows the trend of O–H > N–H > C–H, that is, the O–H bond is most promoted by the oxygen atom. The possible reason is the O–H bond forms the strongest hydrogen bond in the transition state among the X–H bonds investigated in this work. Additionally, it was found that the oxygen coverage has little effect on the X–H bond scission.

 Please wait while we load your content...
Please wait while we load your content...