Halogen-bond driven co-crystallization of potential anti-cancer compounds: a structural study†
Abstract
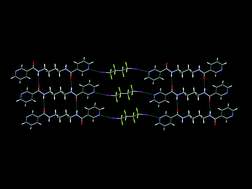
The halogen-bonding ability of a series of bis-pyridine based pharmaceutically active ingredients is explored and the structural behaviour of co-crystals thereof is compared with that of their hydrogen-bonded analogues. All co-crystals are analysed using IR spectroscopy and thirteen new crystal structures comprising iodoperfluoroalkanes and bis-pyridineacetamides are presented. Binding preferences, structural patterns and correlations between molecular and crystal structures are discussed in the context of both hydrogen- and halogen bonds.

 Please wait while we load your content...
Please wait while we load your content...