Three mixed-ligand coordination networks modulated by flexible N-donor ligands: syntheses, topological structures, and temperature-sensitive luminescence properties†
Abstract
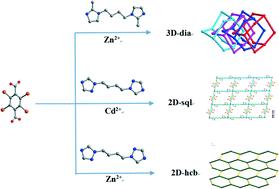
Three new mixed-ligand metal–organic coordination networks based on tetrabromoterephthalic acid and different bis-imidazole or bis-triazole ligands, [Zn2(bmib)(tbtpa)2·2H2O]n (1), [Cd(ditb)(tbtpa)(H2O)2·2H2O]n (2), and [Zn(ditp)(tbtpa)]n (3) (bmib = 1,4-bis(2-methyl-1H-imidazol-1-yl)butane, ditb = 1,4-di(1H-1,2,4-triazol-1-yl)butane, ditp = 1,3-di(1H-1,2,4-triazol-1-yl)propane, H2tbtpa = tetrabromoterephthalic acid), have been synthesized under solvothermal conditions and structurally characterized by single-crystal X-ray diffraction analyses, infrared spectroscopy (IR), elemental analyses, powder X-ray diffraction (PXRD) and thermogravimetric analyses (TGA). Single-crystal X-ray diffraction analysis reveals that 1 features a 4-fold class IIIa interpenetrated dia framework strengthened by Br⋯Br halogen bonds. Complex 2 possesses an infinite 2D layer structure with a 44-sql topology, and the 2D sheets were further packed into the 3D supramolecular framework in an –ABAB– fashion which is reinforced by hydrogen bonds and Br⋯Br halogen bonds. Complex 3 is a 2D 63-hcb network incorporating [Zn2(tbtpa)2] cyclic subunits, and the 2D layers were further extended to a 3D supramolecular framework incorporating π⋯π interactions and Br⋯Br and Br⋯O halogen bonds. In 1–3, all flexible N-donor ligands and dicarboxylates are 2-connected linkers, but networks with diverse topologies are formed, which indicates the coordination preferential geometries of the central metal ion and that the coordination conformations and modes of the ligands have important influences on the resultant structures. Furthermore, there is no solid-state photoluminescence observed for 1–3 at 298 K, and only 2 and 3 are emissive when cooled to liquid nitrogen temperature. The nanosecond range of lifetimes of 2 and 3 in the solid state at 77 K reveals that their emission is fluorescent in nature.


 Please wait while we load your content...
Please wait while we load your content...