Ligand hierarchy on driving the crystal packing. Effect of supramolecular interactions on solid-state conformations adopted by saccharinate Pd(ii) complexes†
Abstract
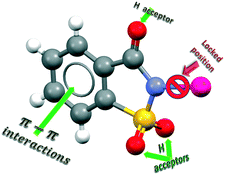
The potential of saccharinate as a supramolecular organizer in selected palladium complexes has been thoroughly evaluated from the perspective of those relevant interactions operating in organic salts and co-crystals in which saccharinate is involved. With this purpose, molecular and supramolecular structures of a series of bis-saccharinate complexes with the general formula trans-[Pd(sac)2(L)2] [L = pyridine (1), pyridazine (2), PPh3 (3), SMe2 (4) or nicotinamide (6)] containing co-ligands with different polarities and abilities to form intermolecular bonds have been studied by X-ray diffraction. These X-ray diffraction data were either previously reported (1 and 3), or obtained from newly determined crystal structures (2, 4 and 6). The new crystal structures of closely related complexes [Pd(sac)2(SMe2)(OH2)]·dmso (5) and [Pd(succinimide)2(nicot)2] (7) have also been elucidated and provide an additional structural discussion. We have disclosed that the anti-configuration is preferred for saccharinate ligands and that the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group defines the most important supramolecular interactions in the crystal packing of 1, 2 and 4. The saccharinate ligand drives crystal packing when pyridine (1), pyridazine (2), PPh3 (3), or SMe2 (4) are the co-ligands, but the crystal packing orienting role relies on water (5) or nicotinamide (6 and 7) when these ligands are present. These results have been compared with other previously reported examples. Molecular geometries have been optimized by DFT, with the interesting output of the energetic preference by the syn-configuration. Supramolecular interactions distort conformations from those optimized ones so that complexes with stronger interacting ligands display larger distortions.
O group defines the most important supramolecular interactions in the crystal packing of 1, 2 and 4. The saccharinate ligand drives crystal packing when pyridine (1), pyridazine (2), PPh3 (3), or SMe2 (4) are the co-ligands, but the crystal packing orienting role relies on water (5) or nicotinamide (6 and 7) when these ligands are present. These results have been compared with other previously reported examples. Molecular geometries have been optimized by DFT, with the interesting output of the energetic preference by the syn-configuration. Supramolecular interactions distort conformations from those optimized ones so that complexes with stronger interacting ligands display larger distortions.

 Please wait while we load your content...
Please wait while we load your content...