Weakening of the π*–π* dimerisation in 1,2,3,5-dithiadiazolyl radicals: structural, EPR, magnetic and computational studies of dichlorophenyl dithiadiazolyls, Cl2C6H3CNSSN†
Abstract
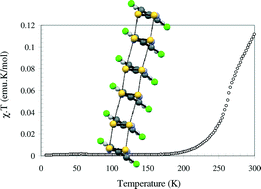
A series of dichlorophenyl dithiadiazolyls (DCP-DTDA) have been prepared and structurally characterized by X-ray diffraction (1 = 2,3-DCP-DTDA; 2 = 2,4-DCP-DTDA; 3 = 2,5-DCP-DTDA, 4 = 3,4-DCP-DTDA and 5 = 3,5-DCP-DTDA). All the radicals adopt cis-cofacial π*–π* dimers with intra-dimer S⋯S contacts in the range 3.119(8)–3.300(5) Å. The propensity for chloro-aromatics to adopt a β-sheet structure assists formation of lamellar structures in 1–5 with intra-stack Cl⋯Cl contacts in the range 3.44(1)–3.82(1) Å. Perpendicular to the stacking direction, the packing is dominated by formation of intermolecular S⋯Cl contacts. Variable-temperature magnetic susceptibility measurements on 1–5 in the range 5–300 K reveal the onset of paramagnetism around ca. 200 K. Solid-state EPR studies reveal that this paramagnetism is associated with thermal population of a triplet excited state. A good fit to the Bleaney–Bowers model determined the exchange interactions (2J) between the radicals in the dimers to be in the range −904 to −1529 cm−1. Computational studies of the dimerisation process indicate that at intra-dimer S⋯S distances in excess of 3.2 Å the closed-shell singlet becomes destabilized with respect to both the open-shell singlet and triplet state configurations.
- This article is part of the themed collection: International Year of Crystallography Celebration: North America

 Please wait while we load your content...
Please wait while we load your content...